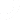One of the most difficult things to understand is the concept of intended use vs. indications for use in the medical industry. These details are very important for people who work in healthcare, the government, and even regular people who want to learn more about medical gadgets and drugs.
Prefer Audiovisuals Over Text? Check This Out!
Understanding the Basics
Intended Use:
At its core, “intended use” means what the company that made a medical product wants it to be used for. This description makes sure that the product meets certain safety and effectiveness standards and is in line with the rules. Not only is the intended use a business statement, but it is also a very important part of regulatory submissions.
For instance, a surgical scalpel intends to make precise cuts during medical treatments.
Indications for Use
But “indications for use” goes into more detail about the specific conditions or situations where a medicine is thought to work well. Additionally, it describes the product’s main features and tells healthcare workers when and how to use it.
A pain reliever, for example, may have indications for use in the treatment of mild to severe pain caused by a number of conditions.
Intended Use vs. Indications for Use – Why Does it Matter?
It’s important to know the difference between “intended use” and “indications for use.” Regulatory agencies such as the FDA carefully scrutinize these aspects to ensure the safety, effectiveness, and proper usage of goods on the market. According to a recent FDA report, a substantial number of product recalls result from incorrect understandings or violations of a product’s intended use and indications for use.
To show how important these differences are, let’s look at a few real-life examples:
Devices for Medicine:
Intended Use: Patients with coronary artery disease are the intended audience for a cardiac stent.
Indications for Use: Healthcare professionals may recommend it for individuals with specific types of artery blockages or those who have recently undergone particular medical treatments.
Pharmaceuticals:
Intended Use: Antibiotics are meant to treat infections caused by germs.
Indications for Use: It may be used to treat skin infections, urinary tract infections, and respiratory illnesses.
Implications for Healthcare Regulatory Compliance:
To get regulatory approval, it is very important to understand and clearly state the planned use and indications for use. Not doing so could push back product sales, recalls, or even legal problems.
Top Tips for Manufacturers:
- Make the intended use clear in product documentation and applications to regulatory bodies.
- To assist healthcare workers, provide clear and precise indications for use.
- Manufacturers should review and update documents regularly to keep up with changing legal standards.
Bridging the Gap for Healthcare Professionals
Getting Healthcare Professionals to Work Together to Improve Patient Safety:
Healthcare professionals are very important for making sure that they use the medical goods as per the direction. It is safer and more effective to care for patients when you understand the differences between intended use and indications for use.
For instance, using a medical gadget incorrectly because you don’t understand what it’s for can cause bad things to happen and put patients at risk.
Educational Initiatives:
Adding thorough training programs for healthcare workers can close the knowledge gap and lower the chance of misunderstandings. Researchers have found that training healthcare workers specifically on using a product in accordance with its intended use and indications for use reduces the likelihood of mistakes.
Conclusion:
Clear language ensures medical goods are safe and effective, transcending mere semantics in healthcare complexities. Clarifying the differences between “intended use” and “indications for use” improves regulatory compliance. This clarity aids healthcare professionals in better understanding, ultimately contributing to enhanced patient outcomes. As we conclude, remember to carefully consider the meaning of each word in the broader healthcare context.